Friday, 13 March, 2026
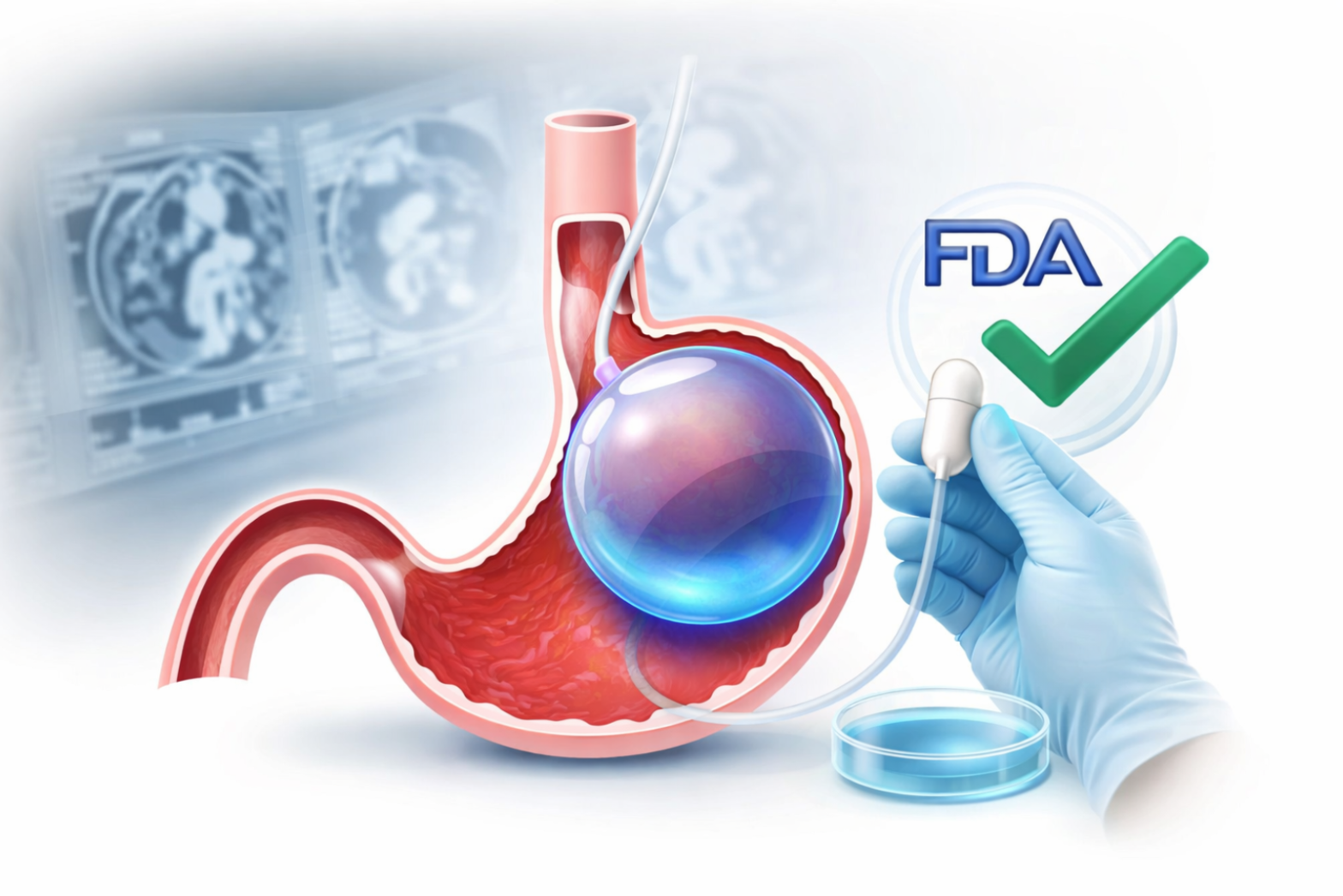
The weight loss industry has entered a transformative phase as the Allurion Balloon receives FDA approval, marking a major milestone in non-surgical obesity treatment. This breakthrough offers patients a procedure-free, anesthesia-free, and endoscopy-free alternative to traditional bariatric options.
For individuals struggling with obesity but hesitant about surgery, this innovation opens a powerful new door.
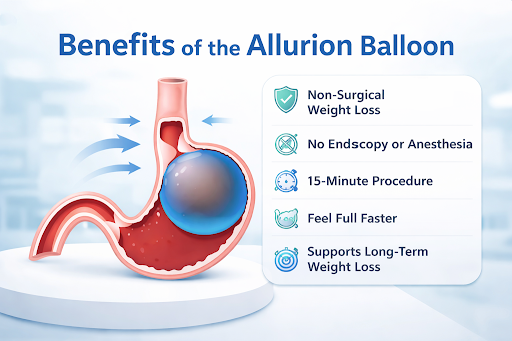
The Allurion Balloon is a swallowable intragastric balloon designed to help patients lose weight without surgery, endoscopy, or anesthesia.
Unlike traditional gastric balloons that require placement and removal through an endoscopic procedure, the Allurion Balloon:
It works by occupying space in the stomach, helping patients feel fuller faster and eat smaller portions.
FDA approval signals that a medical device has met strict standards for:
This approval provides both patients and doctors with greater confidence in the treatment’s credibility and safety profile.
For clinics and weight loss centers, it also means expanded treatment possibilities under regulated medical standards.
Here’s a simplified step-by-step breakdown:
The patient swallows a small capsule attached to a thin catheter.
Once positioned in the stomach, the balloon is filled with liquid through the catheter.
The balloon stays in place for about 16 weeks, helping control hunger and portion Sizes.
The balloon deflates and passes naturally through the digestive system. No surgery. No scars. No anesthesia.
The Allurion Balloon is typically suitable for:
However, proper medical evaluation is always required before treatment.
Patients often experience significant weight loss within the 4-month treatment period
when combined with lifestyle guidance.
Traditional gastric balloons require:
The Allurion Balloon simplifies the entire process, making it more accessible and
less intimidating for patients.
With obesity rates rising globally, many individuals are seeking minimally invasive
solutions that fit into their lifestyle without surgical downtime.
The FDA approval of the Allurion Balloon reinforces a shift toward:
It represents innovation meeting patient comfort.
FDA approval confirms that it meets safety and efficacy standards. However, like any
medical treatment, it should be done under expert supervision.
Weight loss results vary, but many patients lose a significant percentage of excess
weight during the 16-week period when combined with proper dietary guidance
Most patients tolerate the capsule well. Mild discomfort may occur initially as the
body adjusts
No. The balloon stays in the stomach for approximately 16 weeks before passing
naturally.
The FDA approval of the Allurion Balloon marks a major advancement in nonsurgical
weight loss treatments. It provides a safe, effective, and minimally invasive
option for patients who want structured support without going under the knife.
As medical technology evolves, solutions like this are reshaping how we approach
obesity management